Vivalink is a provider of digital healthcare solutions including biometrics technology for virtual patient care and decentralized clinical trials. We leverage unique physiology-optimized medical wearable sensors and data services to enable a deeper and more clinical understanding between provider and patient.

Vivalink introduced eSkin in 2014, which represented the first-of-its-kind breathable thin film substrate with integrated circuits and sensors designed for long term wearable use.
Launched in collaboration with Google ATP as the Digital Tattoo
by Moto X, the technology is now at the core of Vivalink's family of biometric devices and technology.

200+ Commercial Customers
45 Countries Worldwide
21 Research Institutions

Over 100 commercial partners
Reached 30 countries worldwide
Announces Biometrics Data Platform

Announces Large Scale 3,000 Patient AF Trial
ECG Platform Receives FDA Clearance
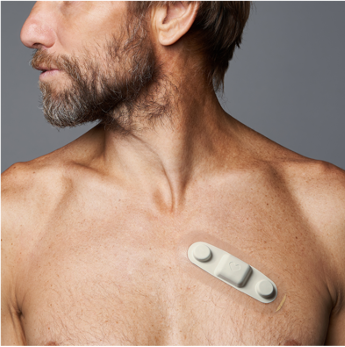
Medical Wearable ECG Sensor Receives CE Mark
Launches Medical Wearable Sensor Platform
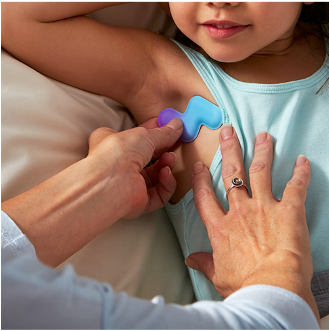
Announces Reckitt Benckiser Partnership
Launches temperature monitor
Developing new products

Starts development of eSkin Technology

Fill out the form below to get in touch with a Vivalink representative.
Talk to a Vivalink expert in connected medical wearables for remote patient monitoring.
With over 100 commercial partners in 25 countries around the world, and a wide variety of experience with patient use cases, Vivalink can help your organization be successful with deploying remote patient monitoring.
Cardiac Arrhythmia
Chemotherapy
Chronic Conditions
Clinical Trials
Coronary Artery Disease
Infection Monitoring
Nursing Home
Post Acute Care
Sleep Bruxism
Stress and Depression
Surgeon Stress
Virtual Hospitals
51 E. Campbell Avenue, Suite 160
Campbell, California 95008
Sales: info@vivalink.com
Support: support@vivalink.com